
Anyone with type 2 diabetes should know that insulin therapy is a must. Insulin is the basic medication, when secretion of insulin from β cells decreases to the extent that it becomes insufficient to maintain normal blood sugar levels. A large clinical study started in 1977 in Great Britain (UKPDS), followed 5102 newly discovered persons with type 2 diabetes during 15 years. It was found that 53% required insulin therapy after 6 years of diabetes. Until the publication of the UKPDS results, insulin was considered the last step in the treatment of type 2 diabetes. Switching to insulin was after 10-15 years of using oral agents, when vascular complications were already advanced. Today, type 2 diabetes is known to be a progressive disease that leads to depletion of pancreatic β cells. Earlier introduction of insulin would slow down that process. It would also improve the response of the liver, muscles, adipose tissue and blood vessels to insulin.
Early insulin therapy reduces the occurrence of vascular complications and weight gain, related to insulin. The introduction of insulin after 5-8 years of diabetes is considered the optimal time, provided that type 2 diabetes is diagnosed on time.

Early insulin introduction naturally follows the changes in T2 diabetes and reduces weight gain and other complications of insulin therapy.
If a person has poorly controlled diabetes for several years, which has led to complications, the introduction of insulin will improve glycosylated hemoglobin (HbA1c), but it will not slow down or prevent the development of new complications. This is important to know! After 12 years of diabetes, a person with retinopathy decides to switch to insulin. He/she has good sugar measurements and better HbA1c, but still doesn’t see!
So, when it comes to insulin – “timing” is important.

When is the optimal time to introduce insulin therapy?
That time is clearly defined.
If a person is on a maximum dose of oral agents and fails to establish morning glucose levels up to 7 mmol/l, as well as HbA1c below 7%, it is time for insulin. There is no difference whether the person is obese or lean.
For most people, this is a period lasting 5 to 10 years after diagnosis of type 2 diabetes.
Then the activity of pancreatic β cells is insufficient to cover the increase in sugar level after a meal, or to keep the sugar normal in the morning, i.e. during the night.
What are the most common fears that patients feel when introducing insulin therapy?
Some fears are real, but some are the result of ignorance.
The real fears are the fear of gaining weight and the fear of severe hypoglycemia. Ignorance leads to the idea that insulin therapy indicates a more severe form of diabetes. Insulin is linked to the need for greater dietary discipline, more selfmanagement and more commitments. However, patients have been shown to have fewer doubts than physicians and are often more insulin-ready than physicians.
What is the expected weight gain on insulin therapy?
It depends on the time of starting the insulin therapy, as well as the type of insulin.
If insulin is introduced early, weight gain is minimal. If HbA1c has been elevated for a long time, the weight gain is greater. Each 1% reduction in HbA1c increases weight for 1.5 to 2 kg. This means that if HbA1c was 12%, normalization of HbA1c with insulin therapy can lead to weight gain of up to 10 kg.
Is insulin given to some people at the time of diagnosis type 2?
Yes.
If there are distinctly elevated glycemia, e.g. over 15 mmol/l with high HbA1c > 10%, insulin should be given immediately after diagnosis. Such therapy will reduce glucose toxicity, which induces β cells’ depletion. Insulin administered at the time of diagnosis of type 2 diabetes can lead to better recovery of β cells. It also reduces metabolic memory – “memory of long-term poor glucose regulation”, that induces tissue glycosylation. After normalization of glycemic values and HbA1c, oral antihyperglycemic agents can be given, and insulin discontinued.

Introduction of insulin after tablet treatment failure
This is no problem and in many health systems, selected physicians introduce insulin. There are printed recommendations how this should be done and it is not “high mathematics”. Everything is simplified precisely because there are many people with diabetes and because it is difficult to get to an internist, and even harder to get to an endocrinologist. The introduction of insulin was facilitated by insulin pens, small needles that do not intimidate and devices for measuring glucose from the finger. As a rule, switching to insulin is performed in an outpatient clinic. The introduction of insulin in a hospital setting is reserved for the elderly, the visually impaired, those with several other diseases and with acute (ketoacidosis) or chronic complications of diabetes (diabetic foot or nephropathy).
Pen insulin complements insufficient insulin secretion from the pancreas. If insulin is started on time, the doses are very small. Basal insulin (Insulatard, Insuman Basal, Humulin NPH) in a dose of 10 units/day or 0.1 – 0.2 unit/kg of body weight (BW) per day is recommended. It is usually started with a dose before dinner or before bedtime. However, if the health insurance allows the start of treatment with an insulin analogue (Levemir, Lantus, Toujeo, Tresiba), the administration time does not have to be in the evening. Older people are often given insulin in the morning to facilitate education. Oral agents are retained and such therapy is popularly called BOT.
BOT = Basal Oral Therapy
If the start of insulin therapy was late, it is necessary to receive insulin twice a day and in a higher dose, e.g. 0.3 – 0.5 units/kg of BW/day. The initial dose is determined by the doctor. However, everyone must be trained to give him/herself insulin, to measure blood glucose levels and to adjust the dose of insulin (a process called titration).
Insulin administration technique
The insulin administration technique involves several things.
If cloudy insulin (mixture or only NPH) is used, it is necessary to shake well the insulin suspension. Many are in a hurry and do not “shake” the pen enough, before giving the insulin. It is necessary to shake the pen 20 times, in order to make a homogeneous suspension. Homogenization is done by rolling the pen between the palms 10 times, after which the pen should be turned up and down, 10 times.
When insulin is injected, the needle needs to stay in the skin for another 10 seconds. This way, the last drop of insulin, which would otherwise leak, is not lost due to surface tension and capillary pressure.
It is necessary to think about the place of insulin administration, having in mind the different blood supply to the skin in different parts of the body. Avoid places with surgical scars and lipodystrophy.
It is recommended that a person does not smoke 30 minutes before and after giving bolus insulin.
Needles
The insulin therapy administered via pen is painless if a single-use insulin needle is used. 1 needle = 1 use. When a new needle is inserted, deep pressure receptors are stimulated, not pain receptors. Each needle has a protective silicone layer that prevents direct contact of the metal with the tissue. However, if one needle is used more than once, the protective silicone layer may peel off, the needle may become dull and thus a feeling of pain may occur.
When a new pen is started, it is necessary to expel the air from the pen. It is usually necessary to squeeze out 4-6 units, in order to get a drop of insulin.
The dose adjustment is simple. Many pens have a magnifying glass to see the numbers. The rotation of the plunger, to determine the dose of insulin, is felt under the fingers, and can also be heard.
Insulin in the pen is given by placing the pen at 90 degrees on the skin. The sting is not felt that way. When the pen is completely on the skin, the insulin injection plunger is pressed.

Self-adjustment or “titration” is simple. It is done according to a “scheme”. If a person takes basal insulin once in the evening, the sugar level is measured every morning. The average of the last 3 measured glycemias on waking is calculated. If the average morning glycemia is > 5.5 mmol/l, the dose of NPH insulin is increased by 2 units. Titration is done weekly. This means that every 7 days the dose of basal insulin can, independently, be increased by the patient by 2 units. For example, if the doctor started insulin with 10 units before dinner, in a month time this can turn into 18 units. If even then the morning glucose levels have not dropped below 5.5 mmol/l, increasing the dose by 2 units per week continues. Of course, nocturnal hypoglycaemia should not occur. If repeated hypos occur, that cannot be explained by any reasonable event, the dose of basal insulin is reduced by 4 units.
The dose of NPH insulin in obese people can be increased by 4 units per week.

Self-adjustment of newer insulin, insulin analogues, is easier, as these analogues “work more stably”. An average of 2 or 3 morning glycemia is always calculated.
Example – corrections for Toujeo insulin:
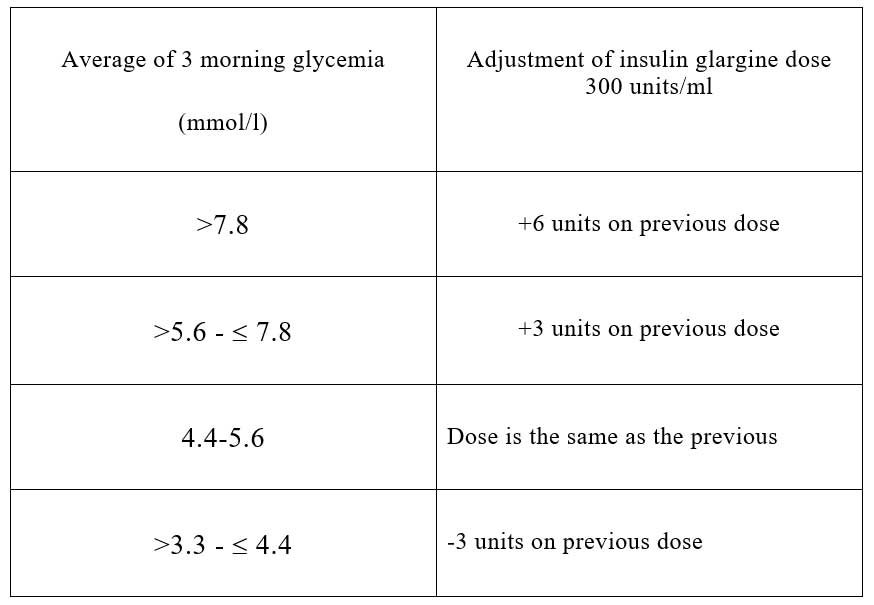
Example – corrections for Tresiba insulin:

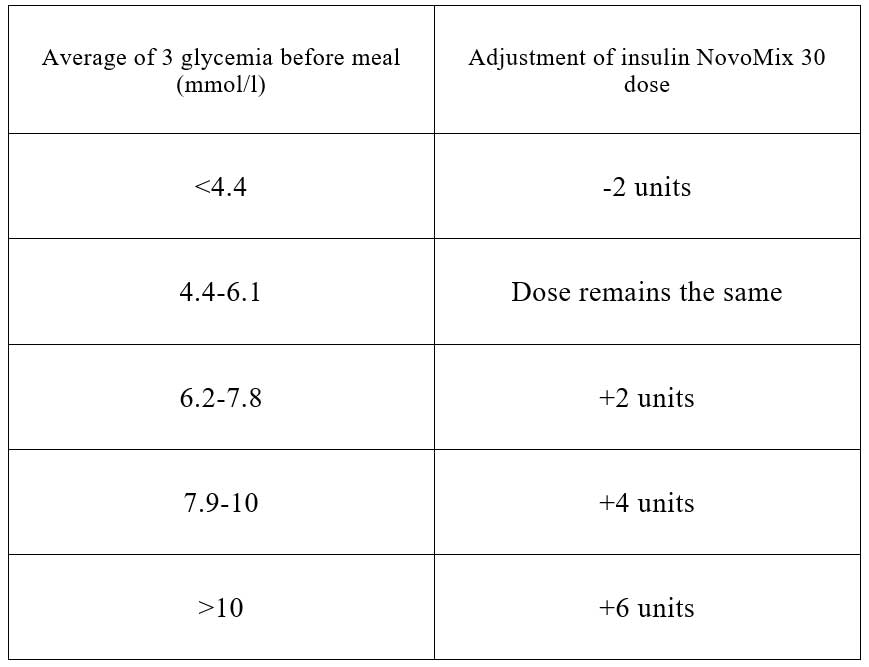
Adjustment for insulin mixtures (Mixtard, Insuman Comb, Humulin M3, NovoMix, Humalog 25 or 50) is a bit more complicated. Titration is done according to the average of 3 morning glycemia, for the evening dose, i.e. the average of 3 glycemia before dinner, for the morning dose.
Example – dose adjustment for NovoMix 30:
Example – dose adjustment for Humalog 50: Adjust the dose before breakfast based on glycemia before dinner. Adjust the pre-dinner dose based on glycemia (on an empty stomach) before breakfast. The change in insulin dose is done the next day.
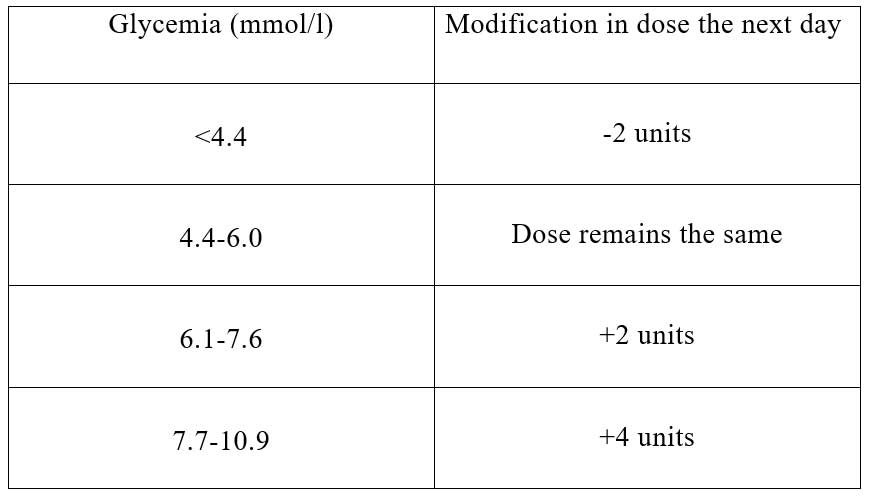
It is necessary to agree with the doctor when self-monitoring is to be done. Each sugar level measurement must be recorded. Initially, the doctor will explain how self-adjustment is done.

The addition of new insulin, usually “short” insulin, to existing insulin therapy is intensification. Example: a person receives 20 units of Lantus (or other basal insulin). HbA1c is over 7%, although morning glycemia targets 4-6 mmol/l are met. What does it mean? It means that blood glucose is elevated during the day – usually after a meal. What is done in that case? Short or “bolus” insulin is added before the biggest meal. There are several “short”insulins :
Actrapid, Insuman Rapid or Humulin R: they start to “work” after 30 minutes and last for 5 hours;
NovoRapid, Apidra, Humalog: they start working after 10 minutes and their effect lasts 3 hours and
FiAsp – it starts working “immediately” and the effect lasts 2 hours.
When a person receives 1 long insulin, which “holds the base” and 1 insulin before the main meal, then such a regimen of insulin therapy is called basal plus.
BASAL PLUS = 1 basal insulin + 1 bolus with a large meal
When a person receives Basal plus insulin therapy, usually with metformin, or other oral agent, and has failed to have HbA1c <7%, then further intensification is performed. With another meal, short insulin is given. It is then said that the person is receiving BASAL 2 Plus therapy. If even such therapy does not give HbA1c <7%, switching to BASAL BOLUS regimen is done. This means that a person receives 1 basal insulin and a bolus (short) insulin before each meal. Most people with type 2 diabetes end up on a basal bolus regimen. This type of insulin therapy is “normal” for type 1 diabetes.


Follow Us